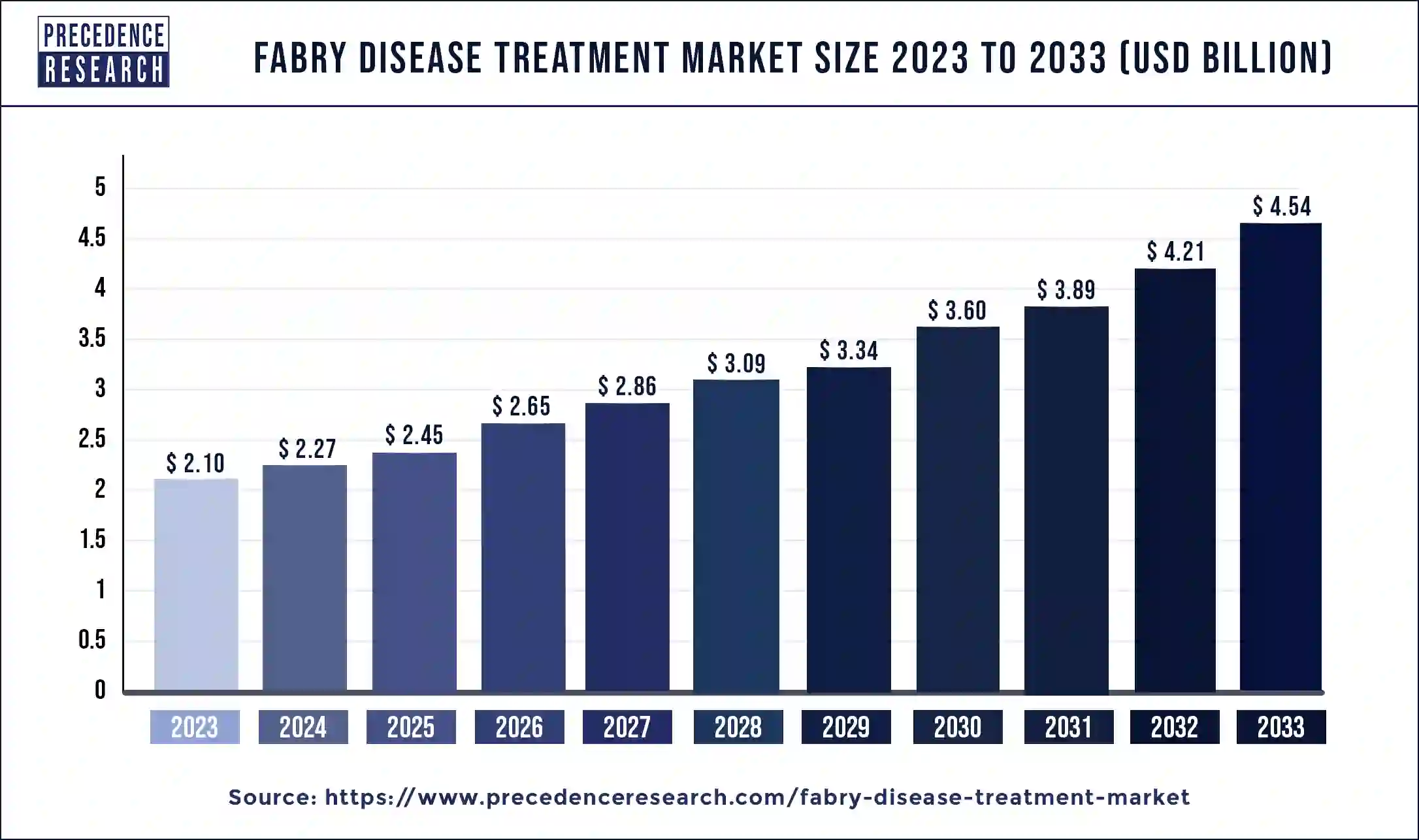
Fabry Disease Treatment Market Size to Worth USD 4.54 Bn by 2033
The global fabry disease treatment market size is expected to increase USD 4.54 billion by 2033 from USD 2.10 billion in 2023 with a CAGR of 8.02% between 2024 and 2033.
Key Points
- The North America fabry disease treatment market size accounted for USD 710 million in 2023 and is expected to attain around USD 1,570 million by 2033, poised to grow at a CAGR of 8.25% between 2024 and 2033.
- North America dominated the market in 2023.
- Asia Pacific is expected to host the fastest-growing market over the forecast period.
- By treatment, the enzyme replacement therapy segment dominated the market in 2023.
- By treatment, the substrate reduction therapy segment grows at a rapid pace in the market over the forecast period.
The Fabry disease treatment market refers to the pharmaceuticals and therapies aimed at managing and alleviating symptoms associated with Fabry disease, a rare genetic disorder characterized by the buildup of a specific type of fat in the body’s cells. The market encompasses a range of treatment options, including enzyme replacement therapy (ERT), substrate reduction therapy (SRT), chaperone therapy, and emerging gene therapies. These treatments aim to address the underlying enzyme deficiency and alleviate symptoms such as pain, kidney dysfunction, and cardiovascular complications.
Get a Sample: https://www.precedenceresearch.com/sample/4404
Growth Factors:
Several factors contribute to the growth of the Fabry disease treatment market. These include increasing awareness about rare diseases among healthcare professionals and patients, advancements in diagnostic techniques leading to early detection of Fabry disease, and ongoing research and development efforts to introduce novel treatment modalities. Additionally, initiatives by governments and non-profit organizations to improve access to treatment for rare diseases contribute to market growth.
Region Insights:
The Fabry disease treatment market exhibits regional variations in terms of prevalence, diagnosis rates, and access to treatment. Developed regions such as North America and Europe typically have higher diagnosis rates and better access to advanced therapies due to well-established healthcare infrastructure and reimbursement policies. However, emerging economies in Asia-Pacific and Latin America are witnessing increasing awareness and efforts to improve access to Fabry disease treatment, presenting opportunities for market expansion in these regions.
Fabry Disease Treatment Market Scope
| Report Coverage | Details |
| Fabry Disease Treatment Market Size in 2023 | USD 2.10 Billion |
| Fabry Disease Treatment Market Size in 2024 | USD 2.27 Billion |
| Fabry Disease Treatment Market Size by 2033 | USD 4.54 Billion |
| Fabry Disease Treatment Market Growth Rate | CAGR of 8.02% from 2024 to 2033 |
| Largest Market | North America |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Fabry Disease Treatment Market Dynamics
Drivers:
Several drivers propel the growth of the Fabry disease treatment market. These include advancements in biotechnology leading to the development of more effective therapies, expanding patient pool due to improved diagnostic capabilities and awareness campaigns, and favorable regulatory frameworks supporting orphan drug development. Moreover, collaborations between pharmaceutical companies, research institutions, and patient advocacy groups contribute to accelerated drug development and market expansion.
Opportunities:
The Fabry disease treatment market presents various opportunities for stakeholders. These include the introduction of novel treatment modalities such as gene therapy and RNA-based therapies targeting the underlying genetic cause of Fabry disease. Additionally, expanding market presence in untapped regions, such as Asia-Pacific and Latin America, offers opportunities for market players to address unmet medical needs and diversify their revenue streams.
Challenges:
Despite the growth prospects, the Fabry disease treatment market faces several challenges. These include the high cost of therapy, which may limit access for patients in certain regions or socioeconomic groups. Additionally, the rarity of Fabry disease poses challenges in conducting clinical trials and obtaining sufficient data to support regulatory approvals for new treatments. Moreover, managing long-term complications and ensuring adherence to treatment regimens remain significant challenges in the management of Fabry disease.
Read Also: Embolic Protection Devices Market Size, Share, Report by 2033
Fabry Disease Treatment Market Recent Developments
- In May 2023, Chiesi Global Rare Diseases and Protalix BioTherapeutics, Inc. received FDA approval for Elfabrio (pegunigalsidase alfa-ix) in the United States for the treatment of adult patients with Fabry disease. Elfabrio is supplied as a preservative-free solution in a single-dose vial.
- In May 2023, Sangamo Therapeutics, Inc., a genomic medicine company, received Fast Track Designation from the FDA for isaralgagene civaparvovec, or ST-920, a wholly owned gene therapy product candidate for the treatment of Fabry disease. ST-920 is currently being evaluated in the Phase 1/2 STAAR study, with a total of 20 patients diagnosed to date.
- In May 2023, Europe allowed Chiesi Farmaceutici and Protalix BioTherapeutics to market their product PRX-102 (pegunigalsidase alfa) in the European territory as a treatment for Fabry disease.
- In September 2022, the agency granted the Orphan Drug Designation (ODD) to AL01211, produced by AceLink Therapeutics, for treating Fabry disease. It is a glucosylceramide synthase (GCS) inhibitor and has shown high potency. The treatment was claimed as a much-needed orally consumed medicine as compared to other available treatments.
Fabry Disease Treatment Market Companies
- JCR Pharmaceuticals
- Sanofi Genzyme
- Green Cross Corporation
- Chiesi Group
- Regenxbio Inc.
- Idorsia Pharmaceuticals
- Protalix BioTherapeutics
- Amicus Therapeutics
Segments Covered in the Report
By Treatment
- Enzyme Replacement Therapy (ERT)
- Chaperone Treatment
- Substrate Reduction Therapy (SRT)
- Others
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/