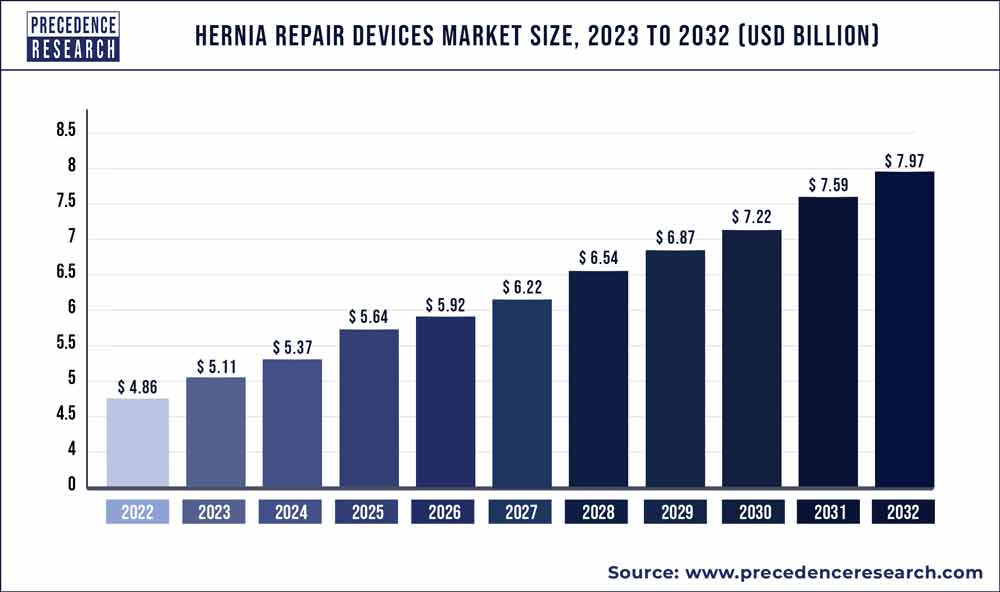
Hernia Repair Devices Market Size To Cross USD 7.97 Bn By 2032
The global hernia repair devices market size accounted for US$ 4.86 Bn in 2022 and is projected to reach around USD 7.97 Bn by 2032, growing at a CAGR of 5.07% from 2023 to 2032.
Report Summary
The global hernia repair devices market report provides a Point-by-Point and In-Depth analysis of global market size, regional and country-level market size, market share, segmentation market growth, competitive landscape, sales analysis, opportunities analysis, strategic market growth analysis, the impact of domestic and global market key players, value chain optimization, trade regulations, recent developments, product launches, area marketplace expanding, and technological innovations.
The study offers a comprehensive analysis on diverse features, including production capacities, demand, product developments, revenue generation, and sales in the hernia repair devices market across the globe.
A comprehensive estimate on the hernia repair devices market has been provided through an optimistic scenario as well as a conservative scenario, taking into account the sales of hernia repair devices during the forecast period. Price point comparison by region with global average price is also considered in the study.
Download Access to a Free Copy of Our Latest Sample Report@ https://www.precedenceresearch.com/sample/2572
Hernia Repair Devices Market Report Scope
| Report Coverage | Details |
| Market Size in 2023 | USD 5.11 Billion |
| Market Size by 2032 | USD 7.97 Billion |
| Growth Rate from 2023 to 2032 | CAGR of 5.07% |
| Base Year | 2022 |
| Forecast Period | 2023 to 2032 |
| Segments Covered | By Product Type, By Surgery Type and By Procedure Type |
Key Highlights:
Reports Coverage: It incorporates key market sections, key makers secured, the extent of items offered in the years considered, worldwide containerized hernia repair devices market and study goals. Moreover, it contacts the division study gave in the report based on the sort of item and applications.
Market Outline: This area stresses the key investigations, market development rate, serious scene, market drivers, patterns, and issues notwithstanding the naturally visible pointers.
Market Production by Region: The report conveys information identified with import and fare, income, creation, and key players of every single local market contemplated are canvassed right now.
Also Read: Cartilage Repair Market Size To Cross USD 4.62 Bn By 2032
Hernia Repair Devices Market Players
The report includes the profiles of key hernia repair devices market companies along with their SWOT analysis and market strategies. In addition, the report focuses on leading industry players with information such as company profiles, components and services offered, financial information, key development in past five years.
Major companies operating in this area
- B Braun Melsungen AG
- Cook Medical
- Medtronic Plc
- W.L. Gore & Associates
- Ethicon Inc.
- C.R Bard Inc.
- Atrium
- LifeCell Corporation
- Baxter International
- Hernia mesh S.R.L.
Market Segmentation
By Product Type
- Hernia Mesh
- Biologic Mesh
- Synthetic Mesh
- Hernia Fixation Devices
- Sutures
- Tack Applicators
- Glue Applicators
By Surgery Type
- Inguinal Hernia
- Umbilical Hernia
- Incisional Hernia
- Femoral Hernia
- Others
By Procedure Type
- Open Surgery
- Laparoscopic Surgery
Regional Segmentation
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Southeast Asia and Rest of APAC)
- Latin America (Brazil and Rest of Latin America)
- Middle East and Africa (GCC, North Africa, South Africa, Rest of MEA)
Research Methodology
Secondary Research
It involves company databases such as Hoover’s: This assists us to recognize financial information, the structure of the market participants and industry’s competitive landscape.
The secondary research sources referred in the process are as follows:
- Governmental bodies, and organizations creating economic policies
- National and international social welfare institutions
- Company websites, financial reports and SEC filings, broker and investor reports
- Related patent and regulatory databases
- Statistical databases and market reports
- Corporate Presentations, news, press release, and specification sheet of Manufacturers
Primary Research
Primary research includes face-to-face interviews, online surveys, and telephonic interviews.
- Means of primary research: Email interactions, telephonic discussions and Questionnaire-based research etc.
- In order to validate our research findings and analysis, we conduct primary interviews of key industry participants. Insights from primary respondents help in validating the secondary research findings. It also develops Research Team’s expertise and market understanding.
TABLE OF CONTENT
Chapter 1. Introduction
1.1. Research Objective
1.2. Scope of the Study
1.3. Definition
Chapter 2. Research Methodology
2.1. Research Approach
2.2. Data Sources
2.3. Assumptions & Limitations
Chapter 3. Executive Summary
3.1. Market Snapshot
Chapter 4. Market Variables and Scope
4.1. Introduction
4.2. Market Classification and Scope
4.3. Industry Value Chain Analysis
4.3.1. Raw Material Procurement Analysis
4.3.2. Sales and Distribution Channel Analysis
4.3.3. Downstream Buyer Analysis
Chapter 5. COVID 19 Impact on Hernia Repair Devices Market
5.1. COVID-19 Landscape: Hernia Repair Devices Industry Impact
5.2. COVID 19 – Impact Assessment for the Industry
5.3. COVID 19 Impact: Global Major Government Policy
5.4. Market Trends and Opportunities in the COVID-19 Landscape
Chapter 6. Market Dynamics Analysis and Trends
6.1. Market Dynamics
6.1.1. Market Drivers
6.1.2. Market Restraints
6.1.3. Market Opportunities
6.2. Porter’s Five Forces Analysis
6.2.1. Bargaining power of suppliers
6.2.2. Bargaining power of buyers
6.2.3. Threat of substitute
6.2.4. Threat of new entrants
6.2.5. Degree of competition
Chapter 7. Competitive Landscape
7.1.1. Company Market Share/Positioning Analysis
7.1.2. Key Strategies Adopted by Players
7.1.3. Vendor Landscape
7.1.3.1. List of Suppliers
7.1.3.2. List of Buyers
Chapter 8. Global Hernia Repair Devices Market, By Product Type
8.1. Hernia Repair Devices Market, by Product Type, 2023-2032
8.1.1 Hernia Mesh
8.1.1.1. Market Revenue and Forecast (2020-2032)
8.1.2. Hernia Fixation Devices
8.1.2.1. Market Revenue and Forecast (2020-2032)
Chapter 9. Global Hernia Repair Devices Market, By Surgery Type
9.1. Hernia Repair Devices Market, by Surgery Type, 2023-2032
9.1.1. Inguinal Hernia
9.1.1.1. Market Revenue and Forecast (2020-2032)
9.1.2. Umbilical Hernia
9.1.2.1. Market Revenue and Forecast (2020-2032)
9.1.3. Incisional Hernia
9.1.3.1. Market Revenue and Forecast (2020-2032)
9.1.4. Femoral Hernia
9.1.4.1. Market Revenue and Forecast (2020-2032)
9.1.5. Others
9.1.5.1. Market Revenue and Forecast (2020-2032)
Chapter 10. Global Hernia Repair Devices Market, By Procedure Type
10.1. Hernia Repair Devices Market, by Procedure Type, 2023-2032
10.1.1. Open Surgery
10.1.1.1. Market Revenue and Forecast (2020-2032)
10.1.2. Laparoscopic Surgery
10.1.2.1. Market Revenue and Forecast (2020-2032)
Chapter 11. Global Hernia Repair Devices Market, Regional Estimates and Trend Forecast
11.1. North America
11.1.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.1.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.1.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.1.4. U.S.
11.1.4.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.1.4.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.1.4.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.1.5. Rest of North America
11.1.5.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.1.5.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.1.5.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.2. Europe
11.2.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.2.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.2.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.2.4. UK
11.2.4.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.2.4.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.2.4.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.2.5. Germany
11.2.5.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.2.5.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.2.5.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.2.6. France
11.2.6.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.2.6.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.2.6.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.2.7. Rest of Europe
11.2.7.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.2.7.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.2.7.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.3. APAC
11.3.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.3.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.3.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.3.4. India
11.3.4.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.3.4.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.3.4.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.3.5. China
11.3.5.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.3.5.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.3.5.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.3.6. Japan
11.3.6.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.3.6.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.3.6.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.3.7. Rest of APAC
11.3.7.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.3.7.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.3.7.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.4. MEA
11.4.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.4.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.4.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.4.4. GCC
11.4.4.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.4.4.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.4.4.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.4.5. North Africa
11.4.5.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.4.5.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.4.5.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.4.6. South Africa
11.4.6.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.4.6.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.4.6.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.4.7. Rest of MEA
11.4.7.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.4.7.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.4.7.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.5. Latin America
11.5.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.5.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.5.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.5.4. Brazil
11.5.4.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.5.4.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.5.4.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
11.5.5. Rest of LATAM
11.5.5.1. Market Revenue and Forecast, by Product Type (2020-2032)
11.5.5.2. Market Revenue and Forecast, by Surgery Type (2020-2032)
11.5.5.3. Market Revenue and Forecast, by Procedure Type (2020-2032)
Chapter 12. Company Profiles
12.1. B Braun Melsungen AG
12.1.1. Company Overview
12.1.2. Product Offerings
12.1.3. Financial Performance
12.1.4. Recent Initiatives
12.2. Cook Medical
12.2.1. Company Overview
12.2.2. Product Offerings
12.2.3. Financial Performance
12.2.4. Recent Initiatives
12.3. Medtronic Plc
12.3.1. Company Overview
12.3.2. Product Offerings
12.3.3. Financial Performance
12.3.4. Recent Initiatives
12.4. W.L. Gore & Associates
12.4.1. Company Overview
12.4.2. Product Offerings
12.4.3. Financial Performance
12.4.4. Recent Initiatives
12.5. Ethicon Inc.
12.5.1. Company Overview
12.5.2. Product Offerings
12.5.3. Financial Performance
12.5.4. Recent Initiatives
12.6. C.R Bard Inc.
12.6.1. Company Overview
12.6.2. Product Offerings
12.6.3. Financial Performance
12.6.4. Recent Initiatives
12.7. Atrium
12.7.1. Company Overview
12.7.2. Product Offerings
12.7.3. Financial Performance
12.7.4. Recent Initiatives
12.8. LifeCell Corporation
12.8.1. Company Overview
12.8.2. Product Offerings
12.8.3. Financial Performance
12.8.4. Recent Initiatives
12.9. Baxter International
12.9.1. Company Overview
12.9.2. Product Offerings
12.9.3. Financial Performance
12.9.4. Recent Initiatives
12.10. Hernia mesh S.R.L.
12.10.1. Company Overview
12.10.2. Product Offerings
12.10.3. Financial Performance
12.10.4. Recent Initiatives
Chapter 13. Research Methodology
13.1. Primary Research
13.2. Secondary Research
13.3. Assumptions
Chapter 14. Appendix
14.1. About Us
14.2. Glossary of Terms
Thanks for reading you can also get individual chapter-wise sections or region-wise report versions such as North America, Europe, or the Asia Pacific.
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com