Medical Device Outsourcing Market Size, Share, Growth, Trends, Report 2027
The medical device outsourcing market is anticipated to garner notable gains, registering a CAGR of 9.98% every year, according to a 2022 study by Precedence Research, the Canada-based market Insight Company.
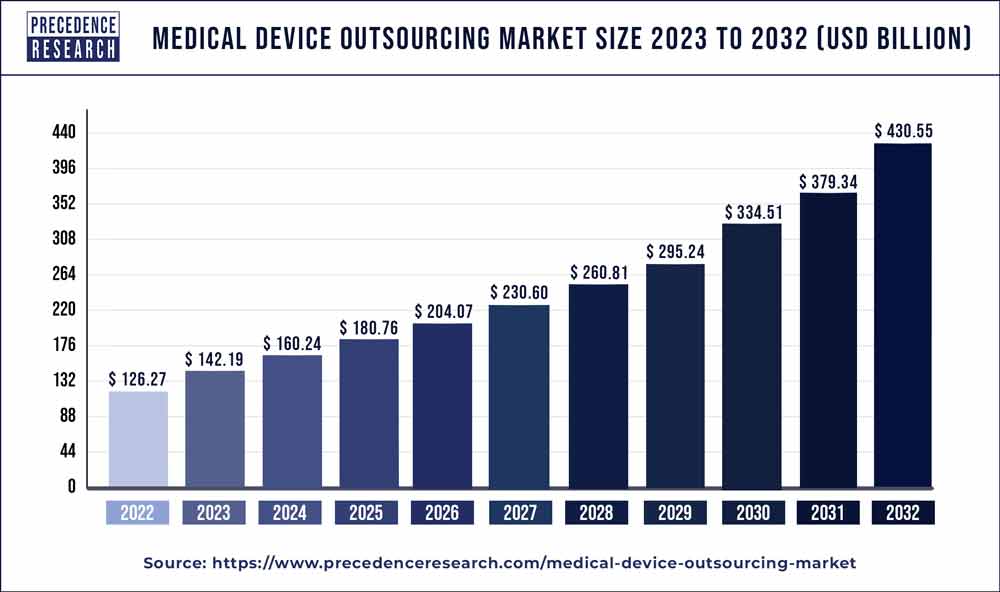
The medical device outsourcing market is surging, with an overall revenue growth expectation of hundreds of billions of dollars within the eight years. The market is expected to grow to US$ 231 billion by 2027. The report contains 150+ pages of detailed analysis.
Medical device outsourcing incorporates the assistance of third party companies for the manufacturing process of medical devices. The shifting trend from in house manufacturing of OEMS towards outsourcing the medical device value chain has fostered the market growth. Additionally, medical devices outsourcing for R&D activities to manufacture quality medical devices is further adding boost to the market growth. Moreover, the growing ageing populations that are more prone to chronic diseases, soaring demand for technological advanced products, stringent regulations for market approval and hasten market entry for novel devices is contributing for the significant market growth. Furthermore, the growing strategic moves such as collaboration and merger & acquisition among CROs and OEMs have supplemented the market for medical device outsourcing. However, the leakage of confidential data is poised to impede the market growth to certain extent.
Our Free Sample Reports Includes:
- In-depth Industry Analysis, Introduction, Overview, and COVID-19 Pandemic Outbreak.
- Impact Analysis 150+ Pages Research Report (Including latest research).
- Provide chapter-wise guidance on request 2021 Updated Regional Analysis with Graphical Representation of Trends, Size, & Share, Including Updated List of figures and tables.
Updated Report Includes Major Market Players with their Sales Volume, Business Strategy and Revenue Analysis by using Precedence Research methodology.
Download a FREE Sample Copy (Including TOC, List of Tables & Figures, and Chart) @ https://www.precedenceresearch.com/sample/1057
Medical Device Outsourcing Market Report Scope
| Report Highlights | Details |
| Market Size | USD 231 Billion by 2027 |
| Growth Rate | CAGR of 9.98% from 2021 to 2027 |
| Largest Market | Asia Pacific |
| Fastest Growing Market | North America |
| Base Year | 2021 |
| Forecast Period | 2021 to 2027 |
Medical Device Outsourcing services as a cost effective curbing tool
Rising awareness of the buyers of medical device industry has enforced manufacturers to adhere various strategies for novel product innovations and administer R&D investments in a cost effective manner. Medical device Original Equipment Manufacturer (OEMs), abruptly adhering to outsourcing services as acost effective curbing tool. Additionally, medical device outsourcing services have benefited the OEMs to reduce R&D and production scale up time as the medical device market has gained high competitiveness in the previous few years.
Regulatory compliance outsourcing is gaining traction in the current market scenario
Medical device manufacturing players are endlessly collaborating with numerous regulatory compliance outsourcing firms to get their devices launched with economical approval in the global market. The outsourcing firms aids in getting quick approval and timely launch of medical devices with several productive benefits including market penetration in emerging countries.
Furthermore, the key characteristics of medical device industry such as rapid time-to-market (TTM), time-to-volume (TTV), and time-to-profit (TTP) are the crucial part of value chain and are fulfilled through third party manufacturing bodies. The OEMs of different companies have adopted the novel strategy to outsource various business prospects such as production, manufacturing, product designing, prototyping and supply chain management among others. This has further helped in reducing the production time and labor cost and the companies can focus on their core skills such as clinical as well as technological advancements.
Read Also: Solid State Battery Market to Record 37.8% CAGR Through 2030
KEY HIGHLIGHTS OF THE STUDY
- The cardiovascular segment is estimated to grow at the remarkable rate during the study period.
- Asia Pacific region is projected to grow at the fastest CAGR during the forecast period owing to the growing spending and up gradation of healthcare industry across the region.
- U.S. dominated the North America Medical Device Outsourcing market and is projected to retain its trend throughout the forecast period.
- Contract Manufacturing of the services segment exhibited major revenue share in the year 2020, whereas, quality assurance service segment poised to grow at the fastest rate during the foresee future.
- In the past one decade, the key medtech companies such as Boston Scientific, Medtronic and Abbott have leveraged the benefit of outsourcing medical device manufacturing process to gain operational efficiencies and market competitiveness.
Asia Pacific exhibited as the most lucrative region in the medical device outsourcing market
Asia Pacific region has outpaced in terms of growth primarily contributed by its key countries such as India and China among others. The emerging countries have contributed immensely for the market growth owing to the high population base, growing demand for single use medical devices, ongoing innovations and technological advancements in medical device industry and increasing chronic diseases have added boost to the market growth. Moreover, the Asia Pacific countries have become attractive outsourcing hubs for medical device manufactures owing to the low operational and labor costs, prominent talent pool and presence of numerous locations for clinical trials.
Some of the noteworthy players in the medical device outsourcing market include:
- Intertek Group PLC
- TüvSüd AG
- Wuxi Apptec
- SGS SA
- Toxikon, INC.
- Eurofins Scientific
- American Preclinical Services
- Sterigenics International LLC
- Pace Analytical Services LLC.
- North American Science Associates, Inc.
- Charles River Laboratories International, Inc.
Market Segmentation
By Service
- Product Upgrade Services
- Regulatory Affairs Services
- Legal representation
- Clinical trials applications
- Regulatory writing and publishing
- Quality Assurance
- Product Maintenance Services
- Product Testing & Sterilization Services
- Product Design and Development Services
- Molding
- Designing & engineering
- Machining
- Packaging
- Product Implementation Services
- Contract Manufacturing
- Accessories manufacturing
- Component manufacturing
- Device manufacturing
- Assembly manufacturing
By Application
- Drug delivery
- Dental
- Diabetes care
- Cardiology
- Endoscopy
- IVD
- Ophthalmic
- Diagnostic imaging
- Orthopedic
- General and plastic surgery
- Others
By Regional
- North America
- U.S.
- Canada
- Europe
- U.K.
- Germany
- France
- Asia Pacific
- China
- India
- Japan
- South Korea
- Middle East & Africa
- Latin America
Thanks for reading you can also get individual chapter-wise sections or region-wise report versions such as North America, Europe, or the Asia Pacific.
Buy this Premium Research Report@ https://www.precedenceresearch.com/checkout/1057
You can place an order or ask any questions, please feel free to contact at sales@precedenceresearch.com | +1 9197 992 333